The evidence
infrastructure for
pain & beyond.
Celéri builds and operates the data backbone that connects point-of-care outcomes, commercial registries, and regulatory-grade real-world evidence — for medtech sponsors, pain physicians, and payers who need proof that works.
App
Enrollment
Direct
of Care
Registry™
Cohort™
Science Cloud
Dashboard
Affairs
Dossier
Ready
Evidence infrastructure.
Not just a registry.
Most evidence programs are built once, for one purpose, and abandoned when the study ends. Celéri builds permanent evidence infrastructure — platforms that generate commercial intelligence today and regulatory-grade data tomorrow, on the same patients, without starting over.
Talk to a StrategistOne platform. Two functions. No rebuild.
Commercial and medical affairs teams draw from the same governed dataset — rep performance and physician-level outcomes for sales, publication-ready longitudinal data for medical. No duplicate infrastructure. No version conflict.
Regulatory-ready from day one.
Every commercial engagement is built on 21 CFR Part 11-compliant, HIPAA-governed infrastructure. When signals confirm the right indication, the evidence base transitions directly into a regulatory-grade cohort — no migration, no lost time.
Point of care to publication.
From a physician's first patient scan to a peer-reviewed manuscript, Celéri's platform spans the full evidence lifecycle — practice registry, sponsored commercial program, regulatory submission, and journal publication — on one connected infrastructure.
One infrastructure. Three audiences.
Celéri's platform is purpose-built for the distinct evidence needs of medtech and pharma sponsors, interventional pain physicians, and payers — each with the view built for their function.
Evidence infrastructure for every stage of growth.
From early R&D feasibility through commercial launch, label expansion, and payer access — Celéri builds the evidence infrastructure that compounds in value at every stage. Deploy a governed real-world evidence registry in 8 weeks. Give your commercial and medical affairs teams real-time outcomes on the same dataset. Activate regulatory-grade evidence when your signals are ready. 85% cost reduction vs. traditional study design.
Explore EaaS™ Platform ›Your practice. Your registry. Your evidence.
PainIntel™ turns every patient encounter into structured, longitudinal outcomes data — automatically, with no staff burden. 30+ validated PRO instruments, 8 EHR integrations at no cost, and a real-time Pain Command Center built for interventional pain, neuromodulation, and pain management specialists. CPT 96127 reimbursement eligible.
Explore PainIntel™ ›Tie reimbursement to what actually works.
Celéri surfaces real-world comparative effectiveness data across pain therapies — what reduces pain, improves function, and cuts downstream utilization costs. Identify overuse, under-response, and true value across your covered population. Evidence in weeks, not years.
Learn More ›A complete evidence program. Delivered as a service.
Building a compliant, commercial-grade outcomes platform in-house requires substantial investment in technology, regulatory expertise, data science, and ongoing operations. Celéri's EaaS model removes those barriers entirely.
Every layer of the evidence stack.
Celéri's products span the full evidence lifecycle — from point-of-care data capture to large-scale real-world benchmarking. Each layer connects to the others.
Celéri EaaS™
End-to-end evidence platform for medtech and pharma sponsors. Commercial Insights Registry™, Regulatory-Grade Evidence Cohort™, Traditional Prospective Study — one governed infrastructure, three evidence tiers.
Learn more › 🩺PainIntel™
Practice-level registry and outcomes platform for interventional pain physicians. 30+ validated PROs, 8 EHR integrations, automated patient outreach, and a real-time Pain Command Center — no staff burden.
Learn more › ☁️PainRWD™ Science Cloud
Large de-identified longitudinal real-world pain dataset — longitudinal PROs, procedures, diagnoses, and utilization signals. Used for external control groups, HEOR modeling, payer dossiers, and benchmarking sponsor evidence.
Learn more › 📱myPainIntel™ Mobile App
Patient-facing mobile app for PRO check-ins, visual pain tracking, and longitudinal outcomes monitoring. Improves completion rates and long-term follow-through in both practice and sponsored registry contexts.
Learn more ›"EaaS is not a software license — it is complete evidence infrastructure, delivered as a service. The clinical story, the commercial intelligence, and the operational scaffolding are all included."
Ready to build your evidence infrastructure?
Whether you're a medtech sponsor at commercial launch, a pain physician building a practice registry, or a payer looking for comparative effectiveness data — Celéri has an evidence pathway for you.
For Med Tech & Pharma
Life science companies – medtech and pharma -seek efficient ways to design studies, collect clinical data, and iterate on innovations. Our platform offers them streamlined research management and connections to qualified investigators. By accelerating evidence generation and providing insights for regulatory and market needs, we help R&D achieve faster, data-driven development.

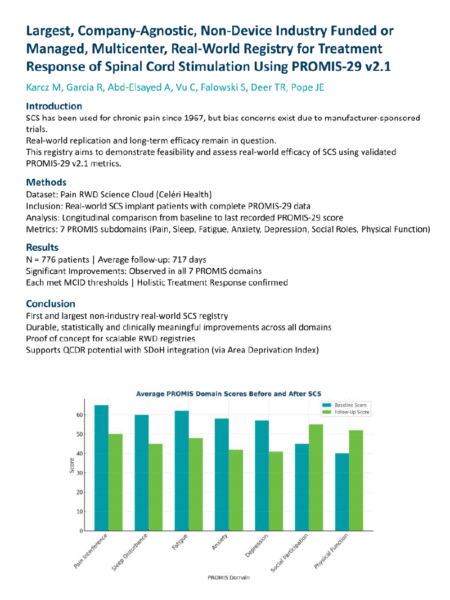
For Clinician-Researchers
Private practitioners have the advantage of agility – they can implement novel techniques or trials faster when not bogged down by academic oversight. Our Research Network physicians report that innovation comes from within the practice and that with the right support they can research, document and scale innovative solutions in community settings. Moreover, clinician-researchers publishing investigator-initiated studies from their own real-world data contribute unique, practice‑grounded insights—expanding product knowledge, uncovering new uses or patient subgroups, and enriching the scientific literature with findings rooted in everyday care contexts.

For Payers
Payers rely on RWE with PROs to fill gaps left by randomized controlled trials, gaining insight into how pain treatments perform across diverse, real-world populations and long-term settings—especially where RCTs may exclude patients with comorbidities or broader demographics. By integrating validated PROs into RWE, payers can better manage utilization, structure value-based contracts, and ultimately ensure treatments deliver real-world outcomes that matter to patients, beyond traditional numeric pain scales.

